
However, Modern Atomic Theory has expanded upon and refined Daltons postulates, incorporating new discoveries and advancements in the field of atomic physics. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms. There is a callout which shows that copper is made up of many sphere-shaped atoms. Daltons Atomic Theory was a significant milestone in the development of atomic theory, providing a basic framework for understanding the behavior of matter. Daltons atomic theory is based on four assumptions Chemical elements are made up of tiny, indivisible, and indestructible particles called atoms. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 2.1.4 2.1.

See how he calculated the mass, volume, and volume of an atom using his model and verified his results with examples. The left stoppered bottle contains copper and oxygen. Learn how John Dalton defined an atom as the smallest indivisible particle and explained its properties using three basic laws of atomic theory. Atomic models have gone through many changes over time, evolving as necessary to fit experimental data. Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. Atoms consist of several types of smaller particles, including protons, neutrons, and electrons. However, scientists now know that atoms are not the smallest particles of matter. Discover what the modern atomic model is called, i.e., 'electron cloud. atomic model, in physics, a model used to describe the structure and makeup of an atom. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. Dalton’s atomic theory was accepted by many scientists almost immediately. (credit copper: modification of work by Copper ). Understand the modern atomic theory and Schrodinger model.
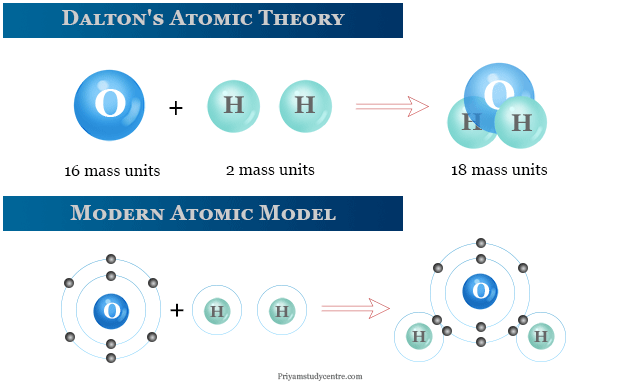
\): When the elements copper (a shiny, red-brown solid, shown here as brown spheres) and oxygen (a clear and colorless gas, shown here as red spheres) react, their atoms rearrange to form a compound containing copper and oxygen (a powdery, black solid).

